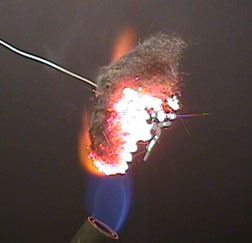
iron + oxygen 
|
|
The iron must be hot and have a high surface area for the reaction to occur this quickly. Iron's common oxidation numbers are +2 and +3. In this reaction it forms a +3 ion when it reacts with a component of the atmosphere.
|
b. Classify the reaction. c. If this reaction occurs much slower in the presence of moisture what do we call it? d. This reaction would go even faster if it were performed in a completely oxygen atmosphere. Why? |