| 1.
| Explain why this statement is true: "If two spots in a chromatogram have the same Rf values, they may be the same compound. If they do not have the same Rf, they are not the same compound."
|
| 2.
| Why is it difficult to separate compounds that have similar structures?
|
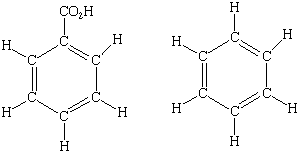
| 3.
| Which of these compounds would have the largest Rf if they were chromatogrammed with a polar stationary phase and a nonpolar mobile phase?
|
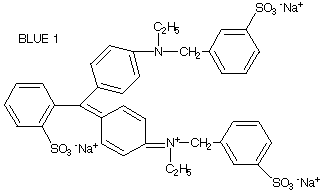
| 4.
| The polarity of the food dyes can be estimated by determining the net charge the dyes would have in solution (note that when the dyes are dissolved in solution, any sodium or hydrogen ions are not attached to the molecule and do not contribute to the net charge).
As an example, Blue 1 has a net charge of -2 (each of the three S03- has a -1 charge; the one N+ has a +1 charge). The larger the net charge, the more polar the dye. Examine the structures of the other dyes and rank them in order from most to least polar.
|
| 5.
| Suppose that the seven FD & C dyes are chromatogrammed using a polar stationary phase and a nonpolar mobile phase. Use the information from question 4 to determine which dye should have the highest Rf and which should have the lowest.
|





