Spectroscopy - Atoms and Light
Energy Levels
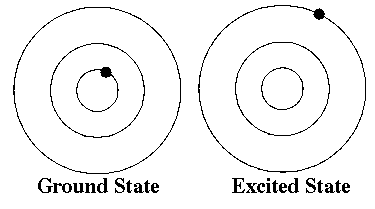
Surrounding a nucleus of an atom are energy levels that the electrons occupy. An electron must reside in one of these levels, not between them. They start filling the lowest energy levels first, those closest to the nucleus, and continue away from the nucleus filling higher energy levels. We often show these energy levels as concentric circles around the nucleus of an atom. This is not a representation of the physical location of the electrons in the atom. It represents the average energy of the electrons in a given level.
When all of the electrons are in the lowest state possible we say that the atom is in its ground state. When they are not in the lowest possible energy level, we say the atom is in an excited state.
Absorption and Emission
An atom changes from a ground state to an excited state by taking on energy from its surroundings in a process called absorption. The electron absorbs the energy and jumps to a higher energy level. In the reverse process, emission, the electron returns to the ground state by releasing the extra energy it absorbed.
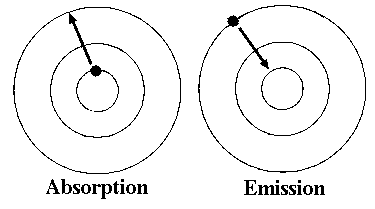
Recall that the electrons must occupy one of the energy levels. To cause a transition between the levels requires an amount of energy exactly equal to the energy difference between the two levels. This has a profound effect on the absorption and emission spectra of an element. Because the atom can only absorb specific amounts of energy, only certain wavelengths of light will be absorbed. Note the two dark lines in the top portion of the figure below. White light was shined on a gaseous sample of atoms. The atoms only absorbed at the two wavelengths (the two dark lines at about 590 nm) because this corresponded to the difference between two energy levels. The result of this absorption was an atom in an excited state.

The excited state atom eventually returns to the ground state. It does this by emitting radiation. The spectrum of the emitted light is shown in the bottom portion of the figure. Note two bright lines appear at exactly the same wavelength as the light that was absorbed.
The most studied atom is the hydrogen atom.







